New research from Emory University indicates that nearly all people hospitalized with COVID-19 develop virus-neutralizing antibodies within six days of testing positive. The findings will be key in helping researchers understand protective immunity against SARS-CoV-2 and in informing vaccine development.
The test that Emory researchers developed also could help determine whether convalescent plasma from COVID-19 survivors can provide immunity to others, and which donors’ plasma should be used.
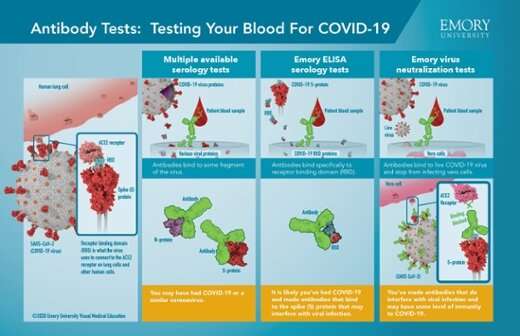
The antibody test developed by Emory and validated with samples from diagnosed patients has demonstrated that not all antibody tests are created equal—and that neutralizing antibodies, which provide immunity, have specific characteristics. Emory’s study focused on those neutralizing antibodies, which can stop the virus from infecting other cells.
The findings are now available on MedRxiv, the preprint server for health sciences, and are not yet peer-reviewed.
In the study, researchers looked at antibodies against the receptor-binding domain (RBD), part of the spike protein on the outside of the virus. The RBD is what grips on to human cells and allows the virus to enter them. The researchers focused on antibodies against the RBD because the sequence of the RBD in SARS-CoV-2 distinguishes it from other coronaviruses that cause the common cold.
The initial 44 patient blood samples used in this study were from patients being treated for COVID-19 at Emory University Hospital and Emory University Hospital Midtown.
“These findings have important implications for our understanding of protective immunity against SARS-CoV-2, the use of immune plasma as a therapy, and the development of much-needed vaccines,” says Mehul S. Suthar, Ph.D., co-lead author and assistant professor of pediatrics at Emory University School of Medicine and Emory Vaccine Center. This study serves as the initial step in a much larger serology effort.
“Very few research teams have done this—looking at neutralizing antibody responses from people who are currently in the hospital,” says Suthar, a virologist and immunologist who specializes in studying immunity to emerging viral infections. “This study provides a snapshot of the immune response as it is happening, not after the battle is over.”
The study’s co-lead author Jens Wrammert, Ph.D., assistant professor of pediatrics at Emory School of Medicine says that the information about RBD-specific antibodies helps inform vaccine development since scientists could test the blood of vaccine study participants for the RBD-specific antibodies, as a gauge of predicted efficacy. It also helps determine the potential best uses of convalescent plasma from the blood of people sickened with COVID-19.
Wrammert says researchers would now use this data to see how they correlate with plasma from convalescent COVID-19 patients. “The fact we are seeing good virus neutralization this early during infection means that we can use binding to the RBD as a way to screen potential plasma donors.”
In addition, Emory researchers were able to rapidly transfer the science to the clinical space and validate the antibody test for clinical, high-throughput use with an additional 231 patient samples from the two hospitals. Led by John Roback, MD, Ph.D., executive vice-chair for clinical operations in the Department of Pathology and Laboratory Medicine and medical director of Emory Medical Laboratories, a multidisciplinary team developed a highly sensitive and accurate test, which ordinarily would take six to nine months, in a matter of weeks.
The Marcus Foundation provided a game-changing contribution toward developing the high-throughput test and was a key factor in how quickly the labs were able to process samples.
“We talk a lot about this ideal situation where you go from the laboratory to actual clinical care,” says Roback. “This is the best example of that taking place in my career. Development of this high-throughput processing has been a tremendous collaboration and monumental effort involving dozens of Emory clinicians, scientists and researchers.”
“The rapid deployment by Emory Medical Laboratories of a validated and highly sensitive assay developed in our laboratory has been truly amazing,” says Wrammert. “This represents a tremendous joint effort among multiple clinicians and scientists at Emory.”
Source: Read Full Article