Many people have experienced infections from E. coli, which are primarily seen as inconvenient and unpleasant. For some patients, like those with blood cancer, however, there is a risk that the bacteria will travel into the bloodstream. In those cases, an E. coli infection is too often fatal. The mortality rate is 15-20%.
The predominant cure for such infections is the use of antibiotics that have detrimental effects on the patient’s microbiome, which play a key part in our physical and emotional well-being, and other side effects. Furthermore, growing problems with antibiotic resistance render such treatments less effective in treating infections.
An international team of scientists has now engineered the first published CRISPR-based candidate for a drug that targets E. coli directly and leaves the microbiome intact. A new paper in Nature Biotechnology titled ‘Engineered phage with antibacterial CRISPR–Cas selectively reduce E. coli burden in mice’ describes the development of the drug candidate to a stage where it is ready for tests on humans.
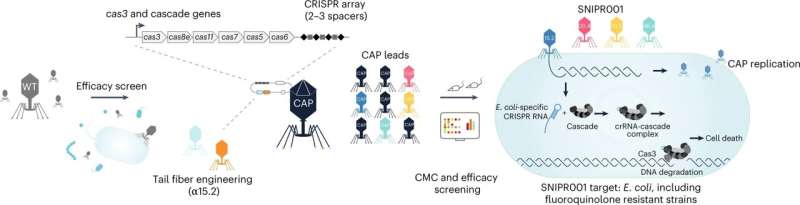
Through extensive use of synthetic biology, the team designed four bacterial viruses that use CRISPR technology to kill the unwanted bacteria precisely.
“We believe that a narrow spectrum drug with these properties could be very useful to cancer patients, among others, who often get serious infections that are difficult to treat with current antibiotics,” says Morten Otto Alexander Sommer, a professor at DTU Biosustain, Co-founder of SNIPR Biome, and lead author of the paper.
The work was carried out in collaboration with JAFRAL (Slovenia), JMI Laboratories (US), and Division of Infectious Diseases at Weill Cornell Medicine (US).
Engineering phages to target E. coli
The team, primarily based at SNIPR Biome, screened a library of 162 naturally occurring phages (viruses that kill specific bacteria). They found that eight of these phages showed promise in targeting E. coli. They then engineered the phages through gene editing to improve their ability to target E. coli.
A cocktail of four of these phages, which they named SNIPR001, very effectively targeted bacteria in biofilms and reduced the number of E. coli in a manner that surpassed that of naturally occurring phages. Further, they showed that the cocktail of phages was tolerated well in the gut of mice and mini pigs while reducing the emergence of E. coli. SNIPR001 is now in clinical development and has been granted a Fast-Track designation (expedited review) by the US Food and Drug Administration.
More information:
Yilmaz Emre Gencay et al, Engineered phage with antibacterial CRISPR–Cas selectively reduce E. coli burden in mice, Nature Biotechnology (2023). DOI: 10.1038/s41587-023-01759-y
Journal information:
Nature Biotechnology
Source: Read Full Article