Researchers have created the largest atlas of post-zygotic genome mutations in healthy human tissue ever assembled—a scientific advancement that could unlock new avenues for diagnosing and treating genetic disease. It is the largest ever in terms of the combined number of tissues and number of donors sampled.
The study, led by researchers at Oregon Health & Science University, published in the journal Science.
The development points the way toward understanding the genetic underpinnings of disease associated with cancer as well as innumerable conditions caused by cellular malfunction, including aging. The atlas could be useful in diagnosing medical conditions, and potentially useful in reversing genetic mutations that cause disease.
“If you’re talking about genetic changes being the basis of disease, there are a wide variety of technologies now that allow us to make changes to the genome,” said senior author Don Conrad, Ph.D., associate professor in the OHSU School of Medicine who directs the Division of Genetics at the Oregon National Primate Research Center. “It may be possible to change those mutations we’ve acquired due to bad luck or bad habits, and change them back to what they were before.”
Window into cell mutations
Researchers generated the atlas using 54 tissue and cell types compiled after death from 948 individuals who donated their bodies for the National Institute of Health’s Genotype-Tissue Expression Program.
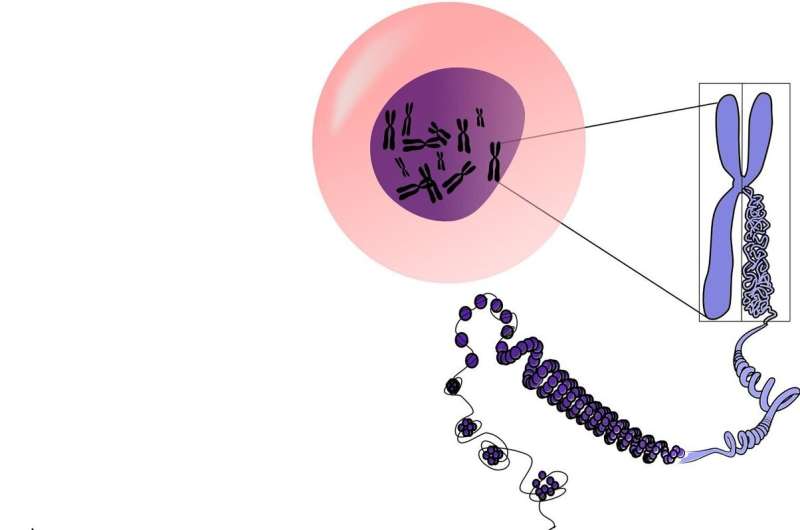
Every person begins as a single cell at the moment of conception, carrying a DNA blueprint within the nucleus of that first fertilized cell. Using those original DNA instructions, the cell divides and replicates into vast groups of cells that form distinct tissues carrying out unique functions in the body. At any one time, the average person is comprised of about 30 trillion cells; in the course of a lifetime, that same person produces quadrillions of cells.
Over time, an individual cell is damaged again and again. In some cases, they repair themselves thousands of times a day.
“Every once in a while, the cell makes a mistake during DNA repair, or misses one—and that’s a change that gets propagated on,” Conrad said. “Our work gives us a window to the extent that those changes occur in different organs and tissues, and during different periods of our lives.”
This situation, known as somatic mosaicism, is a result of cells mutating from the original DNA blueprint.
Building the atlas
Until now, genetic research investigating mutations that occur post-zygotically, or after fertilization, has generally been conducted in biopsies of cancerous tissue such as skin melanomas and lung tumors, or in easily accessible tissues such as blood.
The new atlas opens a field of inquiry into mutations that occur over the course of a lifetime.
“Going from a single cell to a child is a remarkable process,” said lead author Nicole Rockweiler, Ph.D., previously part of Conrad’s lab at OHSU and Washington University in St. Louis, and now a postdoctoral associate at the Broad Institute of Massachusetts Institute of Technology and Harvard University. “When you add on layers of mutations happening at such an important part of our lives, it’s amazing that we can come out pretty well at the end of our gestation.”
To generate the new atlas, researchers developed a computational method using bulk RNA sequencing to characterize the mutations in a massive catalog of tissue samples throughout the body. They were able to trace the point at which mutations occurred by mapping them to a “developmental tree,” indexing them across tissues and among many people.
They found many mutations arose systematically and somewhat predictably as people age, although roughly 10% of mutations appeared to be the result of something intrinsic to an individual, be it genes or environment.
They found most detectable mutations occurred later in life, although many occurred before birth.
“We learned that some tissues, like the esophagus and liver, acquire a lot of mutations whereas other tissues like the brain, acquire fewer mutations,” Rockweiler writes in a post on the Conrad Lab website describing the research. “This made sense to us because the esophagus and liver are exposed to many environmental toxins; here the cells must transmit the message in a noisy environment. A low number of mutations in the brain also makes sense because the brain is primarily composed of cells that don’t replicate.”
More information:
Nicole B. Rockweiler et al, The origins and functional effects of postzygotic mutations throughout the human lifespan, Science (2023). DOI: 10.1126/science.abn7113. www.science.org/doi/10.1126/science.abn7113
Journal information:
Science
Source: Read Full Article